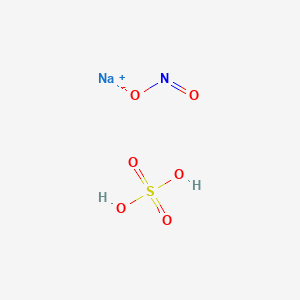
NaNO2/I2 as an alternative reagent for the synthesis of 1,2,3-benzotriazin-4(3H)-ones from 2-aminobenzamides - ScienceDirect

SPECTRUM Sodium Nitrate, Crystal, Reagent, ACS: 7632-00-0, 69, NaNO2, Plastic, Bottle, ACS - 6NNX3|S1355-500GM - Grainger
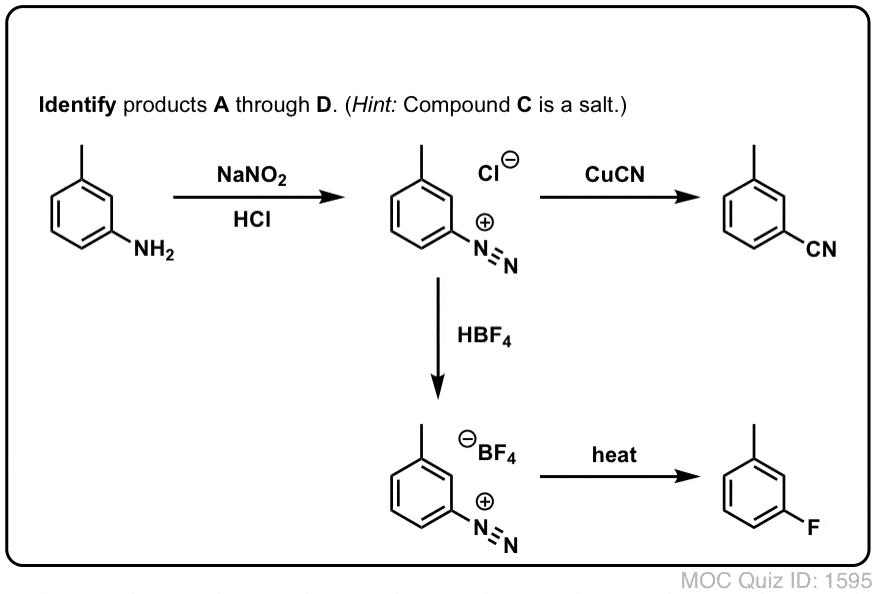
![Compound [A] is an aromatic amine which react with NaNO2 + HCl at 273 - 278 K and form compound [B] . Compound [B] react with HBF4 and the obtain product on Compound [A] is an aromatic amine which react with NaNO2 + HCl at 273 - 278 K and form compound [B] . Compound [B] react with HBF4 and the obtain product on](https://haygot.s3.amazonaws.com/questions/774137_738479_ans_d47545d3f5ad465d9fd600a977e7df56.png)
Compound [A] is an aromatic amine which react with NaNO2 + HCl at 273 - 278 K and form compound [B] . Compound [B] react with HBF4 and the obtain product on

NaNO2/I2 as an alternative reagent for the synthesis of 1,2,3-benzotriazin-4(3H)-ones from 2-aminobenzamides - ScienceDirect

Organic Solid Acid/NaNO2: An Efficient System for the Oxidation of Urazoles and Bis-Urazoles under Mild and Heterogeneous Conditions

Applications of Sodium Nitrite in Organic Synthesis - Mukhopadhyay - 2019 - European Journal of Organic Chemistry - Wiley Online Library

Molecules | Free Full-Text | Silica Sulfuric Acid/ NaNO2 as a Novel Heterogeneous System for the Nitration of Phenols under Mild Conditions | HTML

Reagents and conditions: (a) NaNO 2 , CH 3 COOH, 0°C, 24 h, 90%; (b)... | Download Scientific Diagram

Scheme 1. Reagents and conditions: (a) (i) NaNO2, HCl, 0 °C, 30 min;... | Download Scientific Diagram

Applications of Sodium Nitrite in Organic Synthesis - Mukhopadhyay - 2019 - European Journal of Organic Chemistry - Wiley Online Library
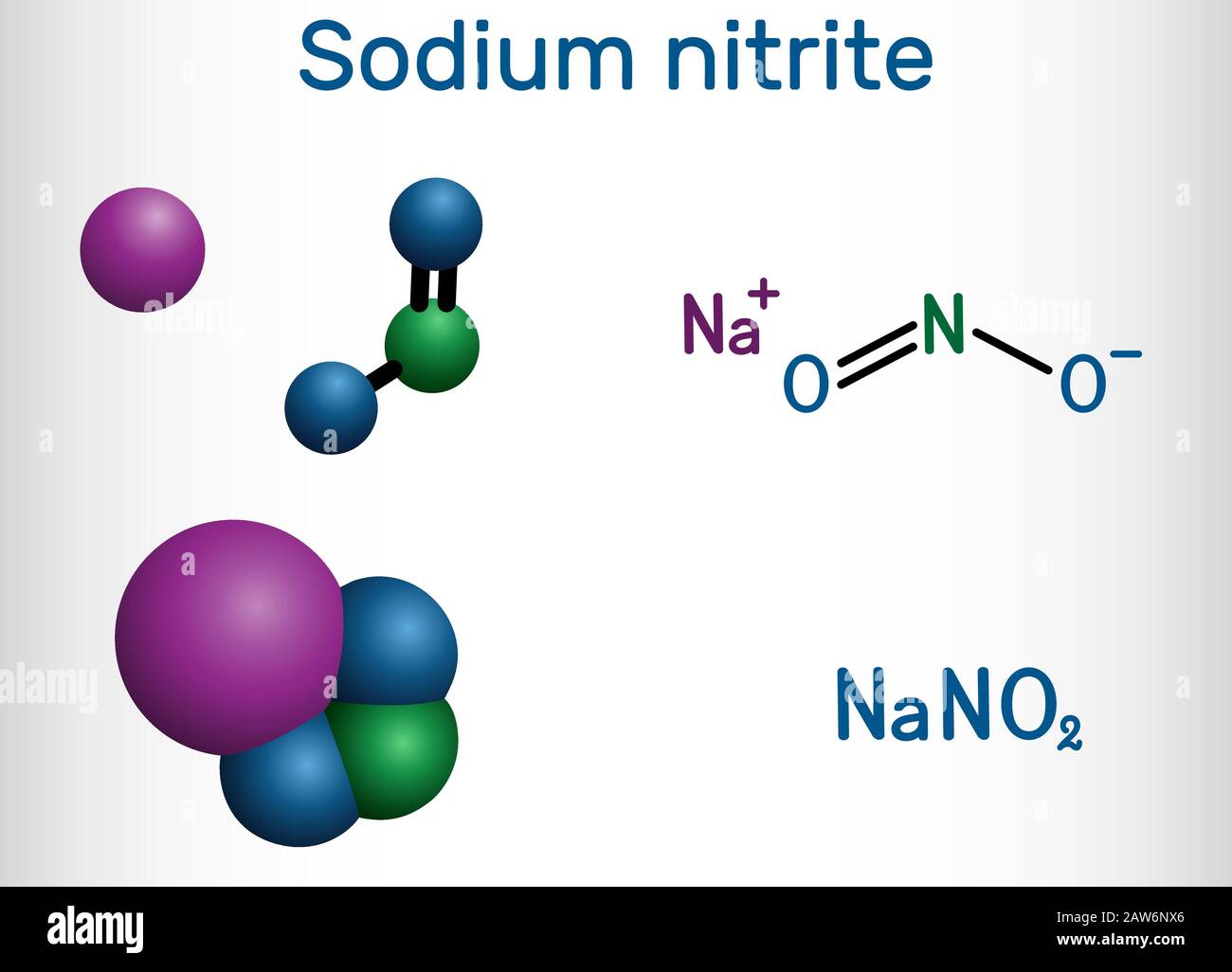
Sodium nitrite, NaNO2 molecule. It is used as a food preservative and antidote to cyanide poisoning. Structural chemical formula and molecule model. V Stock Vector Image & Art - Alamy